An Update On Drugs In Development For COVID-19
By John Cahill

If you work in the regulatory, R&D, or manufacturing areas of drug development, you’ve likely been reading about the many scientific and technical advances that are being fast tracked at present in response to the current COVID-19 global emergency. Since you’re familiar with the lengthy and expensive process of drug discovery, testing, and trials, it’s also likely you know the speed and urgency at which our industry is moving to develop an effective vaccine and treatment is something to be proud of. It is now understood by researchers, companies, and regulators that while ensuring safety, efficacy, and quality is still of critical importance — speed is now paramount! Here are some examples of candidates that are being developed at breakneck speed.
- The drug Favilavi, which was the first approved coronavirus drug in China, has reportedly shown efficacy in treating COVID-19 safely in a clinical trial involving 70 patients.
- The first COVID-19 vaccine in China is expected to be ready for clinical trials by the end of April. Inovio Pharmaceuticals plans to begin human clinical trials on their coronavirus vaccine INO-4800 in April this year. Results from the clinical trials are expected to be available in September 2020. Inovio aims to produce one million doses of the vaccine by the end of 2020 to perform additional clinical trials and/or emergency use of the vaccine.
- Regeneron has accelerated its timeline for having doses of a potential vaccine (REGN3048 and REGN3051) and treatment ready for human clinical trials by early summer. Large-scale manufacturing could start by the middle of April.
- Gilead’s Remdesivir (GS-5734), an experimental broad-spectrum antiviral, originally designed to treat Ebola, has undergone clinical trials in China. The trials are being performed on 761 patients in a randomized, placebo-controlled, double-blind study at multiple hospitals in Wuhan. The results from the trials are expected to be available over the next few weeks.
- Moderna’s mRNA-1273 vaccine will undergo a Phase 1 clinical study in April with support from the NIH, and the company is already preparing for a Phase 2 study. The vaccine targets the Spike (S) protein of the coronavirus.
- Roche’s Actemra has been approved in China for the treatment of severe complications related to coronavirus. Actemra is being evaluated in a clinical trial in China, which is expected to enroll 188 coronavirus patients. The clinical trial is expected to be conducted until May 10.
- Many clinical trials are underway with Chloroquine for fighting COVID-19. This drug has been shown to be safe and efficacious against malaria for decades. Coronavirus patients administered with the Chloroquine Phosphate drug achieved a better drop in fever and shorter recovery time in clinical trials being conducted in Chinese hospitals. Large scale clinical trials with this drug are imminent.
- An intranasal Covid-19 vaccine (similar to NasoVAX) is being developed by U.S.-based clinical-stage biopharmaceutical company, Altimmune. Animal studies are being progressed.
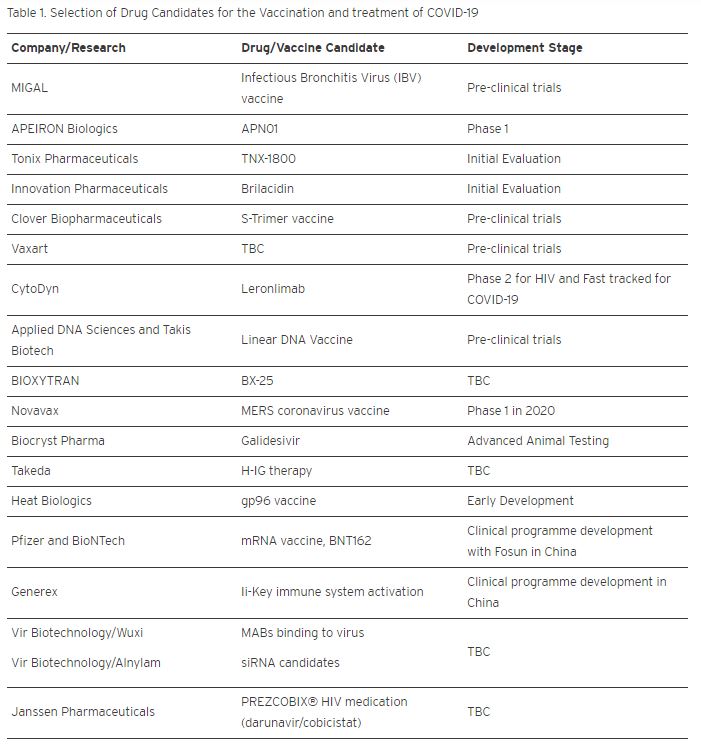
Table 1 below summarizes the many other candidates at different stages of the condensed drug development lifecycle.
As evident above, drug discovery and clinical research activities in COVID-19 prevention and treatment are increasing in pace. It would not be surprising if a workable vaccine has already been discovered and is being tested at present. It will take some time to establish the complete safety and efficacy of the successful candidates, but with the combined knowledge and urgency that now exists in R&D, we should have reason to be confident of a future without COVID-19. In the meantime — and unfortunately this may still be some months away — we must follow national health authority guidance, trust in science, and stay safe for each other.
John Cahill is director, senior consultant, quality and compliance at PharmaLex Ireland.