Bio/Pharma Industry Improves Downstream Operations
By Eric Langer, President and Managing Partner, BioPlan Associates, Inc.
Best Practices: Membrane Technology Options
 Downstream purification includes multiple steps, such as intermediate and polishing chromatography and virus filtration. Many of those steps involve drug products at their most valuable — where a lot of work has already been done and a misstep can risk millions of dollars. So a lot of interest is paid to efficient operations. And as upstream productivity increases, running the downstream side of facilities without creating capacity problems has become a key concern. Challenging aspects of downstream operations include cost of chromatography materials, lack of single-use (disposable) options, cost of membranes, and cleaning and validation costs. Avoiding the high cost of Protein A affinity resins is a goal, but most biomanufacturers are reluctant to make any changes to existing processes, and there are, as yet, few alternatives proven at a larger scale.
Downstream purification includes multiple steps, such as intermediate and polishing chromatography and virus filtration. Many of those steps involve drug products at their most valuable — where a lot of work has already been done and a misstep can risk millions of dollars. So a lot of interest is paid to efficient operations. And as upstream productivity increases, running the downstream side of facilities without creating capacity problems has become a key concern. Challenging aspects of downstream operations include cost of chromatography materials, lack of single-use (disposable) options, cost of membranes, and cleaning and validation costs. Avoiding the high cost of Protein A affinity resins is a goal, but most biomanufacturers are reluctant to make any changes to existing processes, and there are, as yet, few alternatives proven at a larger scale.
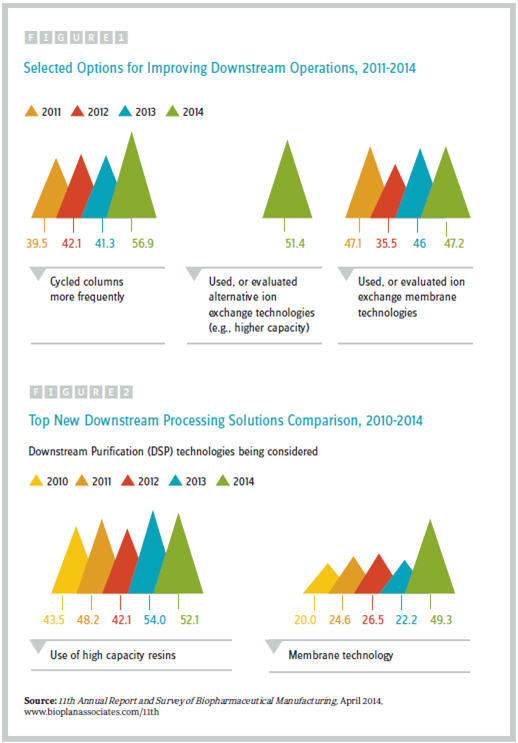
As results from our 11th Annual Report and Survey of Biopharmaceutical Manufacturing (see www.bioplanassociates.com/11th) attest, industry suppliers and end users are developing and evaluating new technologies for improving their downstream processes. As part of our annual survey, we evaluated factors that have led to improvements in operations as well as to the new technologies under consideration.
Cycling Columns Vs. Investigating New Technologies
Our study reveals that 57 percent of respondents cycled columns more frequently last year to improve their downstream purification operations; that’s a big increase from recent years (~40 percent from 2011 through 2013). Ion exchange technologies are also being investigated to a significant degree:
- Slightly more than half of the industry used or evaluated alternative ion exchange technologies, such as higher capacity; and
- Slightly fewer than half used or evaluated ion exchange membrane technologies. (See figure 1)
Results from our study indicate that a significant proportion of industry respondents report changing buffer volumes, actively identifying/assessing bottleneck points, and investigated alternatives to protein A.
As in recent years, though, interest in Protein A alternatives is far greater than actual implementation. Although 3 in 10 purported to have investigated alternatives, fewer than 7 percent claim to have actually made the switch to using alternatives.
For nearly every implementation factor, CMOs report higher levels of adoption, with this likely a reflection of their dependence on efficiency, their managing multiple projects and high turnover, and their ability to pass the costs of new technology implementation onto developers. Indeed, the data is clear that CMOs lead developers in implementing new downstream technologies.
Protein A alternatives provide a good case in point: While biotherapeutic developers were almost twice as likely as CMOs to say they had investigated alternatives to protein A, CMOs were nearly four times as likely as developers to say they had actually implemented alternatives. The results suggest that the technology evaluation process is lengthier for developers than for CMOs, who are more willing to try out new technologies on the basis of the factors outlined above.
Membrane Technology Takes A Leap
In addition to evaluating implementation of downstream technologies, our study looked at the new or alternative technologies respondents are considering currently by asking respondents, “Which new downstream purification (DSP) technologies are you actively considering to address bottlenecks and problems?” Note, this only asked about “actively considering,” indicative of potential future adoption, with this not including those already having adopted these technologies and those considering, but not “actively” pursuing this interest.
On this front, while the use of high-capacity resins was again the most widely-considered technology, the largest industry shift was toward membrane technology. This year, 49 percent of respondents indicated that they are actively considering membrane technology, roughly double the proportion observed in recent years (20-26.5 percent from 2010 through 2012). Consideration of membrane technology was high among both U.S. and Western European respondents and was a top-two consideration for both developers and CMOs.
CMOs showed greater levels of interest in most technologies identified in the study — prepacked columns (64 percent of CMOs versus 30 percent of developers) and use of filters instead of resin chromatography (45 percent of CMOs versus 23 percent of developers). Some of the largest discrepancies occurred in regard to single-use products.
There were some areas, though, in which developers showed keener interest:
- online analytical and control devices
- centrifugation
- development of mAB (monoclonal antibody) fragments
- 2-phase systems.
A small proportion of developers is also considering countercurrent chromatography and field fractionation, with these technologies not generating any interest from CMO respondents.
Interestingly, respondents in Western Europe outpaced those in the U.S. in consideration of the majority of technologies, with some notable exceptions being for disposable UF (ultra filtration) systems, in-line buffer dilution systems, and centrifugation.
Solutions Are Still Forthcoming
Our survey data continue to show that U.S. and European biomanufacturing facilities are considering — but slowly adopting — improvements and new downstream technologies. Overall, CMOs, not developers, are taking the lead in adopting new or streamlined DSP approaches, with CMOs much more motivated by cost savings and associated needs to develop and adopt standardized manufacturing platforms.
The survey captures continued concerns regarding the limitations of existing facilities to keep up with the relatively rapid increases in titer from upstream processes. There is no reason to think that this issue will go away. Upstream process development will continue to improve, and facilities that have a fixed DSP capability will therefore continue to push their DSP to feasible limits.
Survey Methodology: The 2014 Eleventh Annual Report and Survey of Biopharmaceutical Manufacturing Capacity and Production yields a composite view and trend analysis from 238 responsible individuals at biopharmaceutical manufacturers and contract manufacturing organizations (CMOs) in 31 countries. The methodology also included over 173 direct suppliers of materials, services, and equipment to this industry. This year’s study covers such issues as: new product needs, facility budget changes, current capacity, future capacity constraints, expansions, use of disposables, trends and budgets in disposables, trends in downstream purifi cation, quality management and control, hiring issues, and employment. The quantitative trend analysis provides details and comparisons of production by biotherapeutic developers and CMOs. It also evaluates trends over time and assesses differences in the world’s major markets in the U.S. and Europe.
