Digital Ledger Technologies Supporting Regulatory Exchange
By Brooke Casselberry
 In 2016, Don and Alex Tapscott published the book,Blockchain Revolution — How The Technology Behind Bitcoin Is Changing Money, Business, And The World, defining blockchain technology as a protocol for data control and distribution while providing the essential, yet missing, quality of trust, accountability, and integrity of both data and consumers in a distributed ledger environment.
In 2016, Don and Alex Tapscott published the book,Blockchain Revolution — How The Technology Behind Bitcoin Is Changing Money, Business, And The World, defining blockchain technology as a protocol for data control and distribution while providing the essential, yet missing, quality of trust, accountability, and integrity of both data and consumers in a distributed ledger environment.
Much of the book and the initial application of blockchain technology is focused on how it can improve finance, government control (e.g., voting), art and music, and a number of other industries that could be clearly improved through trusted protocol exchange. One industry, however, that has been slightly undervalued is regulatory in life sciences.
Ironically, at the same time that the Tapscott brothers were generating excitement about a technology that clearly has the potential to improve everything it touches (depending on who’s speaking about it), another “revolution” started taking root in regulatory with a larger focus on data.
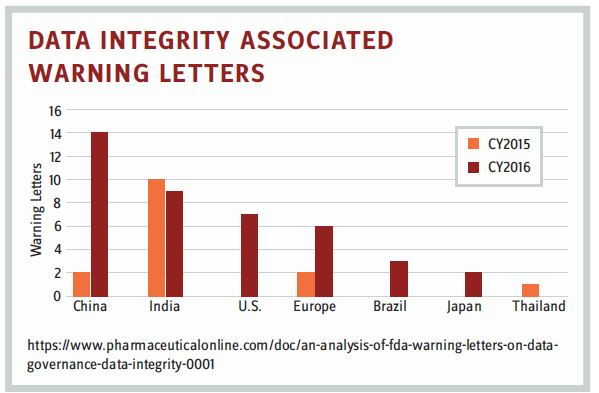
2016 was a year riddled with data-focused initiatives from health authorities and life sciences sponsors. The FDA released a draft guidance document on data integrity and GMP practices defining data integrity and expectations for data compliance in order to avoid being issued a Form 483 for cited failures. In December 2018, the FDA updated this guidance, “Data Integrity and Compliance with Drug cGMP Questions and Answers Guidance for Industry,” further clarifying the agency’s view on data control, trust, and accountability. Warning letters for data integrity findings during GMP inspections increased substantially between 2015 and 2016, identifying a lack of data control within global manufacturing sites. Because blockchain technology is developed using SHA-256 (an algorithm designed by the NSA) cryptography, it can be used to capture, store, and version documents or data. It is immutable and secure, and the content defined on the chain can be accessed only through permission or consensus as defined for purpose. For example, multichain technology provides control of data streams through timestamping, encryption, permissioned distribution, and verification connecting multiple chains of data. This type of data control and security provides data integrity as defined in the FDA cGMP guidance for industry:
- backup data is exact and complete
- stored to prevent deterioration or loss
- documented at the time of performance
- records retention
- complete records of data
- review and validation of accuracy.
The Center for Devices and Radiological Health (CDRH) released a draft guidance, “Use of Real-World Evidence to Support Regulatory Decision–Making for Medical Devices,” on July 27, 2016, detailing the agency’s position on real-world data (RWD) and real-world evidence (RWE) to enhance decision making and identification of device safety and quality in noncontrolled environments such as in-home use. The concept of RWE/ RWD carries over into life sciences decision making for product development, marketing, expanded patient population, pharmacovigilance, and safety.
However, the data needed to provide true evidence of real-world interactions comes from multiple places and has multiple owners. Getting control of the data into centralized repositories or data lakes, or even having simple access to the data, in some cases, is impossible. One ex ample is personal data regulations to protect individual content such as General Data Protection Regulations (GDPR). Through decentralized and/or distributed blockchain technologies, it is possible to access data from multiple sources and distribute data or data bits through validated transactions based on defined consensus. For example, digital currency such as bitcoin operates using this model for distribution of coins through networks without discrimination. The users are protected and secure, and currency is key encrypted, with only the owner in possession of the key. With the ability to consume data from multiple sources (e.g., EHRs, insurance claims, clinical trials, observational studies, mobile devices, safety registries, and patient interactions), metrics and KPIs can be evaluated and incorporated into future product development and business strategies.
In 2017, the Federal Notice (FN) on the “Draft Standardization of Pharmaceutical Quality/Chemistry Manufacturing and Control (PQ/CMC) Data Elements and Terminologies” was published by the FDA and HHS for public comment. The FN continues to build on the data-centric model, identifying the intent of standards for specific elements within PQ/CMC content for the product development life cycle.
While the health authorities will ultimately define the standards for both the data and method of exchange, there is an opportunity for industry to identify its own method of data exchange. For example, the data supporting the development of a product across the life cycle is not only shared with the health authorities for approval, but it is also shared with partners, country affiliates, investors, and other invested parties. Implementing industry standards for collaboration with external sources where agreements of exchange, compromise, and promises are validated and released through protocol using blockchain technology is possible. Platforms and technologies that support this message exchange include Hyperledger, Ripple, and other Ethereum-based smart-contract capabilities.
The biopharma industry continues to invest in innovation to improve both data control and management of the product development life cycle. In a typical regulatory environment, content supporting a product from investigation to post-marketing approval and registration in a global landscape can be contained in anywhere from one to many systems. This can result in a series of challenges, including:
- data and content duplication
- the intent of purpose/definition could vary
- the quality of the data could be questioned
- it adds complexity to the reuse and distribution of the content
- during a merger or acquisition, it could be more difficult to harmonize these data sources with the acquiring company’s system.
Not unlike the cGMP data integrity scenario provided, the need to get control of data is applicable across the complete product life cycle and well into post-marketing activities. Additionally, as the data revolution continues, the traditional sense of separating the life cycle activities from pre-approval to post-approval activities becomes outdated; real-world evidence must be collected at both stages for true analysis.
According to The Pistoia Alliance, 60 percent of pharmaceutical and life sciences professionals are either using or experimenting with blockchain today, compared to 22 percent when asked in 2017. Gartner estimates that by 2030, the business value for blockchain technology will reach $3 trillion, with 10 to 20 percent of the global economic infrastructure running on blockchain.
The life sciences industry is entering a data revolution, a revolution that is inevitable and otherwise semi-forced by the technological advancements over the past 20 years. The ability to garner command and control of dispersed data with various owners and intended purposes is possible for the first time through blockchain technology and coupled with the functions of AI and robotic processing automation (RPA). Life sciences will be able to expedite patients’ accessibility to products, enhance safety, and improve decision making for both business and individual needs. Understanding the regulatory landscape, both past and future, and the drive toward data transformation and standards, provide the framework for the implementation of emerging technologies and overall environmental improvements.
BROOKE CASSELBERRY, MSRA has been a regulatory consultant for 20 years focusing on data, technologies, small business entrepreneurship, and regulatory compliance. She is currently director consulting expert with CGI Technologies for R&D and technology innovation.
