Finding The Right CRO For Rare Disease Trials
By Ed Miseta, Chief Editor, Clinical Leader
CRO selection is a challenge for every company outsourcing clinical trials. When the trial you are about to launch is for a rare genetic disease and has 30 patients spread across several countries and continents, the challenges are multiplied.
This was the situation for Cerenis Therapeutics, which recently launched a Phase 3 trial for patients having very low levels of high-density lipoprotein (HDL), also known as good cholesterol. The low levels of HDL make patients prone to cardiovascular events such as stroke or heart attack. The Cerenis medicine, CER-001, attempts to mimic HDL, allowing the body to rid itself of cholesterol.
In this Q&A, Cerenis founder and CEO Jean-Louis Dasseux discusses the challenges of rare disease trial and why the selection of the right CRO is so important to the success of the trial.
ED MISETA: In rare disease trials, does the CRO selection process become more difficult?
 JEAN-LOUIS DASSEUX: I think so. Since this is the first time a trial is addressing a rare ailment, there is really no CRO on the planet with experience dealing with this patient population. That means the experience is generally coming from the sponsor. Therefore, the role of the CRO becomes more critical. They have to be the liaison between the pharma company and the sites and patients and are responsible for transferring that experience from one group to the other. They also have to be experts in the conduct of a trial. It’s a lot of responsibility for them.
JEAN-LOUIS DASSEUX: I think so. Since this is the first time a trial is addressing a rare ailment, there is really no CRO on the planet with experience dealing with this patient population. That means the experience is generally coming from the sponsor. Therefore, the role of the CRO becomes more critical. They have to be the liaison between the pharma company and the sites and patients and are responsible for transferring that experience from one group to the other. They also have to be experts in the conduct of a trial. It’s a lot of responsibility for them.
MISETA: Have there been any situations where you requested that someone at the CRO be removed from a study?
DASSEUX: That was necessary once, and we felt the individual at the CRO was simply not the right person for the trial. In that case, the concern was the individual’s technical knowledge of trials and how to handle a study where good clinical practice and adhering to requirements set by regulators was critical. But passion is also something we look for in a CRO.
We need to have emotion, to have people who are dedicated to the study. Above all else, this is a human adventure. What is more important to me than the size of the CRO is choosing the one we feel will be most engaged in the study. Some of the larger CROs are not interested in a study involving 30 patients. As a result, we have struggled to find appropriate CROs and will often look for ones that are local to the areas where we are conducting the studies. I believe interactions with the CRO are essential to this relationship, and we are deeply involved with them. We will generally have interactions with them on a daily basis.
MISETA: When you believe a personnel change is necessary, that must be a difficult conversation. Is it usually done at a high level?
DASSEUX: Yes. We try to be a responsible company, and we certainly don’t want anyone to take something like that personally. What we will try to do is go to upper management within the company and let them know we believe an issue exists with one person involved in the trial, or that we feel there is a dedication issue. I tell them that a different type of person might be more appropriate and ask if they can resolve it internally.
We are certainly not looking to be confrontational in any way. If we think a change is necessary, we want to handle it in the most delicate manner possible. In a rare disease trial, we expect a great deal of commitment from all of our partners. That often includes a large time commitment on the part of key individuals. These are all good people, but sometimes that required commitment is just not possible due to personal reasons or family needs.
In those cases, we will reach out to the person to see if they can perform better. If we still see some issues, then we will go to upper management at the CRO to see if they can help us find a solution.
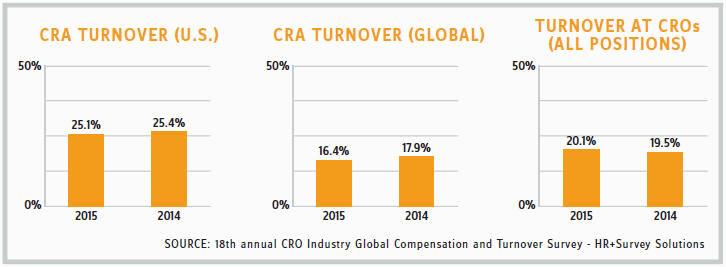
MISETA: Many sponsors also complain about personnel changes at the CRO. Is this ever an issue for you?
DASSEUX: Yes, definitely. I think it is a problem for the entire industry. You have one team leader at the beginning of a study, and then three months later you are working with someone else. We all know this is due to the pressure and challenges these folks face, and the fact is they are always jumping from one CRO to another. It’s a difficult situation for everyone. We will always explain to the CRO the difficult nature of rare disease trials and will request that they assign us someone who is stable and able to stick with the trial until the very end. We always stress the importance of that relationship, because we feel it is critical to the trial.
MISETA: Everyone always wants the CRO’s “A-Team,” but we know it is not possible for every company to get it.
DASSEUX: Exactly. And not being a Big Pharma company, we do not have the deep pockets that allow us to always get our way. We also do not have a huge internal team that can help with the transition from one team leader to another. For that reason we have to be deeply involved in all of the activities being handled by the CRO and have regular meetings with them. That is just an additional cost of running a rare disease indication clinical trial.
MISETA: Are there rewards to that type of model as well?
DASSEUX: Yes. The other side of the coin is there are many CROs that share our philosophy. We now have many of them calling us and saying they would love to work with our company because of these relationships we develop. The value we get from these human relationships will definitely work both ways.
The ideal CRO for us is an organization that is passionate about the disease and excited to be working on a trial that has the potential to become a breakthrough therapy that will save the lives of patients. With many CROs, we find they value in working closely with our team and not some faceless department within a much bigger organization.
MISETA: Do you find there is an appreciation on the part of the CRO when they feel you are treating them the same way you want to be treated?
DASSEUX: Yes, and that is a key point. At the end of the day we are all people, and drug discovery is definitely a human adventure, as I mentioned earlier. Every little step we take has a big impact on the overall success of the trial. This includes patients being motivated and not being made to feel they are guinea pigs. Physicians have to understand the value the trial brings to their patients. Then consider all the other partners involved — the CROs, suppliers, carriers, and those teams involved in all of the different countries. We all have to be on the same page, working together, and personally involved with the effort. If one of those links fails, the trial will not be a success. Everyone has to be committed to working together as a team.
