Influence Of Size In CRO Selection
By Rebecca McAvoy

It may seem as though there are countless factors to consider when selecting a CRO. Don’t get me wrong; there certainly is a lot to consider. For that reason, ISR provides data that can be used to prioritize what is important in the provider selection process. We surveyed hundreds of clinical development outsourcers about what they consider important when selecting providers. To capture the increasing role that preferred provider agreements are playing in the selection process, respondents were asked about attribute importance in several scenarios: 1) choosing among preferred providers, 2) choosing a provider not on their preferred list, and 3) choosing a provider in the absence of a preferred list.
Capturing these nuances affords those working at sponsor organizations the opportunity to directly apply attribute-importance data from the appropriate decision- making scenario to the selection qualifications employed at their companies. Those from CROs can use the information to walk into a bid defense in any of the decision-making scenarios armed with a solid sense of what the industry feels is important when selecting a provider in that situation.
PROVIDER SIZE & THE EFFECT ON THERAPEUTIC EXPERTISE
Therapeutic expertise has remained among the most important attributes from year to year, across trial phases, regardless of whether there are preferred provider agreements in place. An organization’s therapeutic expertise (or lack thereof) is often commented upon when respondents explain their reasoning for assigning overall satisfaction scores for CROs they have recently used. In short, a provider’s knowledge of the desired therapeutic areas and its prior experience working in these areas go a long way in determining whether a provider is chosen for a clinical study.
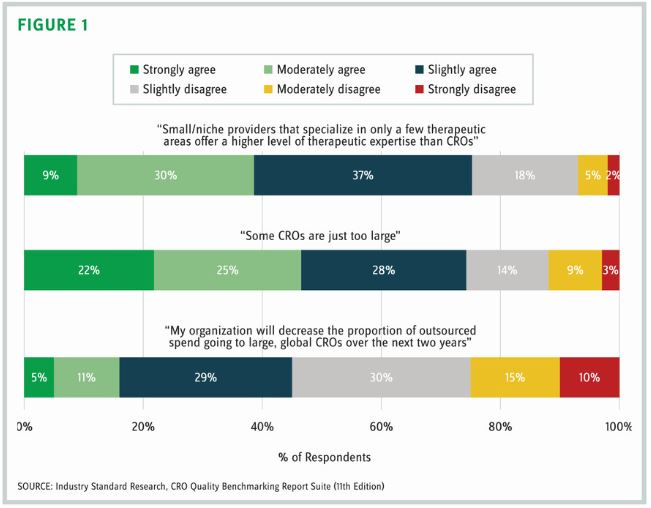
Where does CRO size come into play? Size undoubtedly is a factor in an organization’s ability to deliver global studies or offer a one-stop-shop experience. But how does provider size affect customers’ perceptions of therapeutic expertise? Over the last decade of researching service provider selection, ISR often has seen outsourcers attributing smaller CROs with superior therapeutic expertise in their areas of specialty compared to large CROs, and we wanted to gather the opinions of a larger group. We asked over 500 clinical development outsourcers about their levels of agreement with the statement: Small/niche providers that specialize in only a few therapeutic areas offer a higher level of therapeutic expertise than large CROs (figure 1).
Turns out, about three-quarters of respondents agreed with this statement. About 40 percent indicate that their agreement is moderate to strong. Unquestionably, there are many other factors that go into the decision of which CRO will shepherd a molecule through critical clinical trials — geographic footprint, service capabilities, project manager quality, patient recruitment abilities, and cost, just to name a few. However, the perception that small/niche providers have better therapeutic expertise than their larger counterparts is an interesting one. Small organizations can tout their deep therapeutic expertise in specific areas in marketing messaging, and this data shows that audiences are likely to find these claims credible. Large CROs may do well to message around their therapeutic expertise as well — perhaps along the lines of “Our therapeutic breadth doesn’t come at the expense of depth.”
Respondents were also asked to rate their agreement with the statement: Some CROS are just too large (figure 1). Again, three-quarters of respondents agreed that some CROs are just too large. And, again, this is a sentiment seen in open-ended responses over the years, often referencing cumbersome processes and poor communication in larger CROs.
The following quotes are from 2018 and 2019 studies regarding reasoning for recent user satisfaction ratings for large CROs: “[Large provider is] too big of an organization to get anything effectively done.” “Their way of working, due to [being a] big organization, is sometimes lagging when rapid decisions [are] required.” “They are becoming too large of a CRO.” “Too big [of a] CRO for Phase 1.” “Sometimes it feels too big to be as responsive as needed.” “Too large [of an] organization, bureaucracy overload.”
WHAT’S NEXT?
So where does all this leave us? ISR asked respondents to rate their agreement with a third statement regarding CRO size: My organization will decrease the proportion of outsourced spend going to large, global CROs over the next two years (figure 1). In this case, responses were more split. Nearly half (45 percent) agreed that they would decrease the proportion of spend going to large CROs over the coming two years.
Although respondents didn’t have a unified voice about their companies’ expected future usage of large CROs, this is a statistic to take note of. If nearly half of outsourcers expect usage of large CROs to decline, this spells opportunity for smaller CROs, which should consider heralding their small size and deep therapeutic expertise as benefits. Large CROs may want to consider proactively marketing around attributes traditionally considered strengths of smaller CROs, such as flexibility.
REBECCA MCAVOY is VP of market research at Industry Standard Research.
Survey Methodology: Industry Standard Research is a full-service market research provider to the pharma and pharma services industries. ISR’s CRO Quality Benchmarking research is conducted annually via an online survey. For the 2019 CRO Awards data, more than 60 service providers were evaluated on over 20 different performance metrics. Research participants were recruited from biopharmaceutical and medical device companies of all sizes and are screened for decision-making influence and authority when it comes to working with CROs. Respondents only evaluate companies with which they have worked on an outsourced project within the past 18 months. This level of qualification ensures that quality ratings come from actual involvement with a business and that companies identified as leaders are backed by experiential data.
